Mostly all materials, if not already liquids, can be converted to liquids by digestion methods (e.g., dissolving in acid) allowing for routine elemental analyses. Atoms of the samples emit characteristic light while residing in the torch, which is detected and quantified by the spectrometer.
In the case of inductively coupled plasma (ICP) excitation-based AES, liquid samples are atomized into an extremely fine mist and aspirated into a plasma torch. To excite these species, such that they will emit sufficient light to allow for routine detection, samples are most commonly exposed to plasma. As the name implies, it relies on the detection of light emitted by atoms, and very simple compounds (oxides, nitrides, etc.), of excited atoms/simple compounds. Wolfgong, in Handbook of Materials Failure Analysis with Case Studies from the Aerospace and Automotive Industries, 2016 3.1 Atomic Emission SpectroscopyĪES sometimes referred to by the acronym OES (optical emission spectroscopy), is an elemental analysis technique with detection limits of subparts per billion for most elements. If available use the spectroscope on any vapour lamps which may be available, and compare their spectra.William J.Repeat steps 2-6 with the salts of at least two other.Dip the cleaned nichrome wire into the mush of the salt to be tested and place the end of the wire into a blue flame and observe with the spectroscope.Add a small amount of the salt to be tested and mix with a little hydrochloric acid on a watch Be careful here – use just enough of the acid to give you a semi-solid ‘mush’ of crystals.You may have to repeat this step several times, especially towards the end of the experiment. Dip the flame test wire into concentrated hydrochloric acid, then hold it in the hottest part of the Repeat the process until there is little or no colour from the flame test wire in the flame.
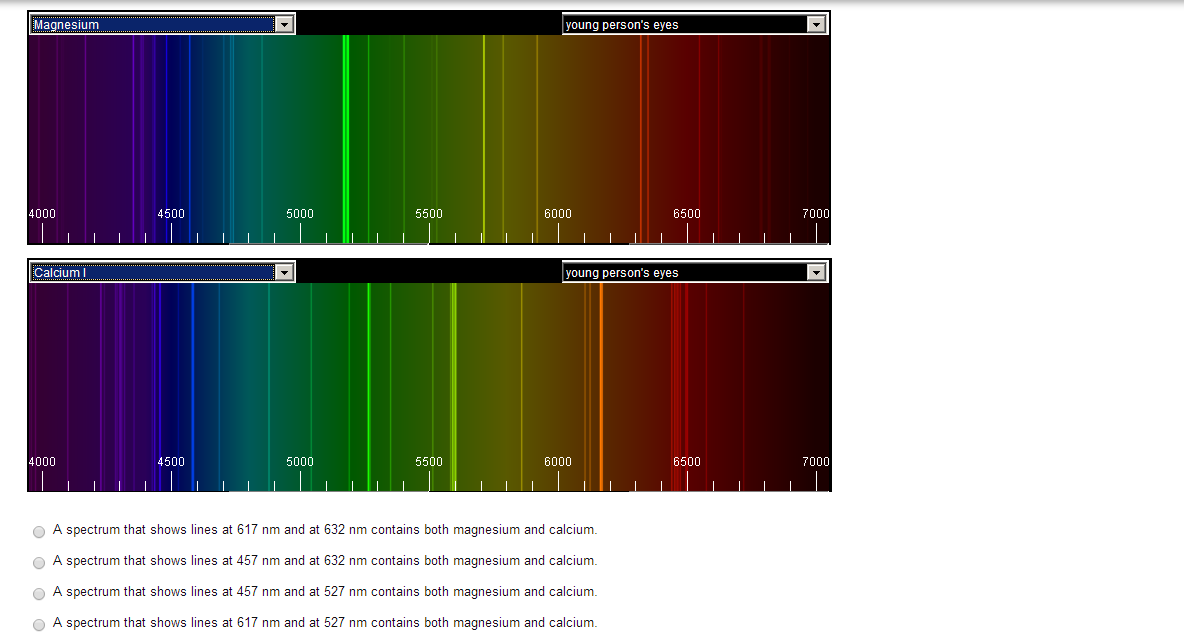
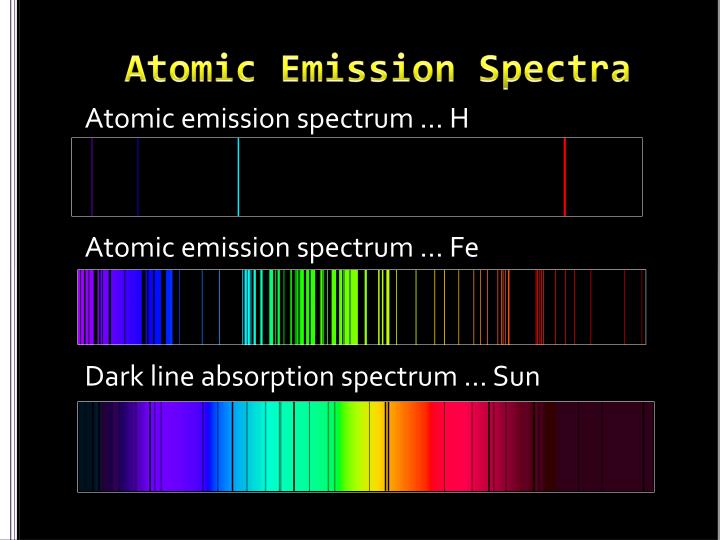
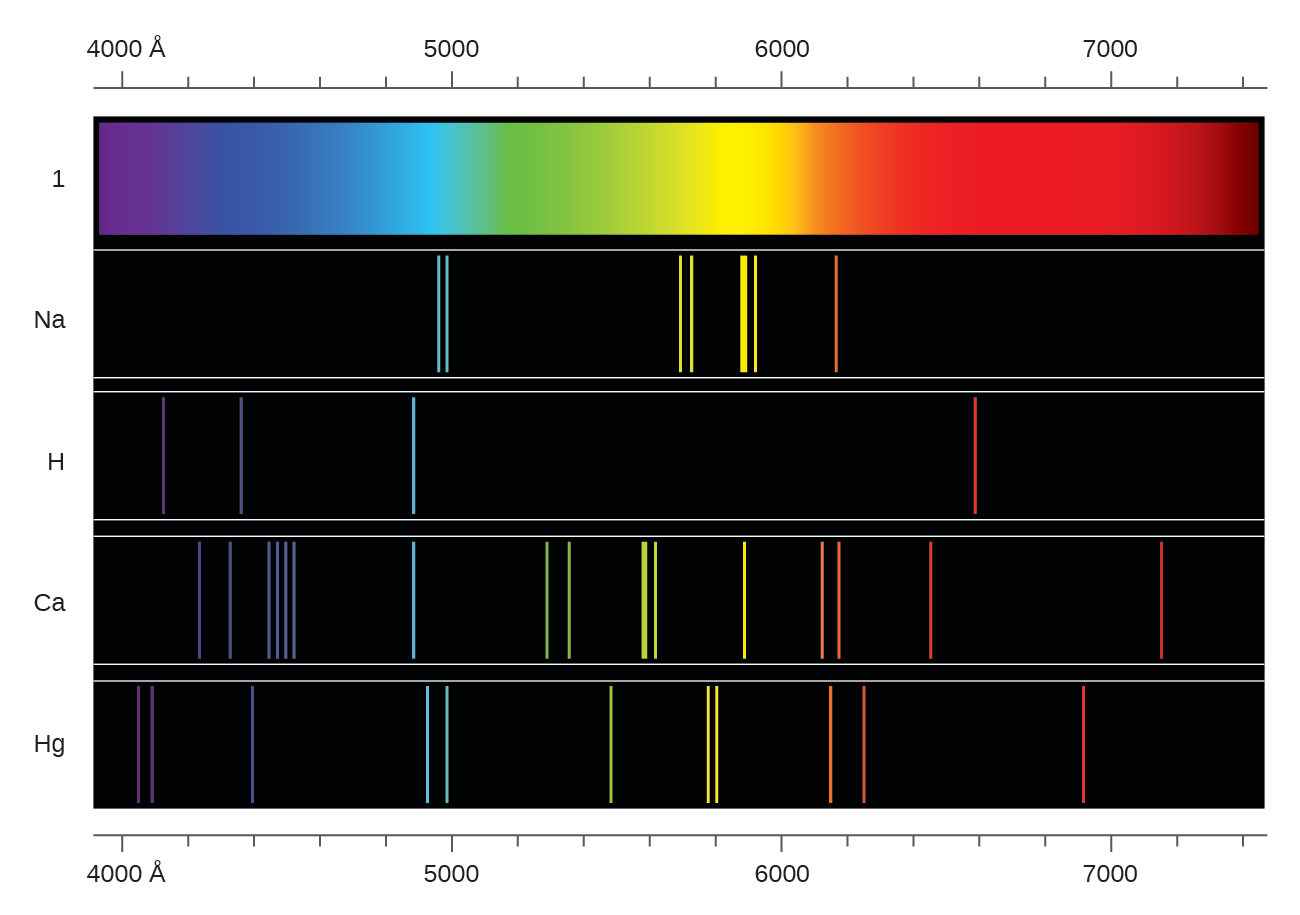
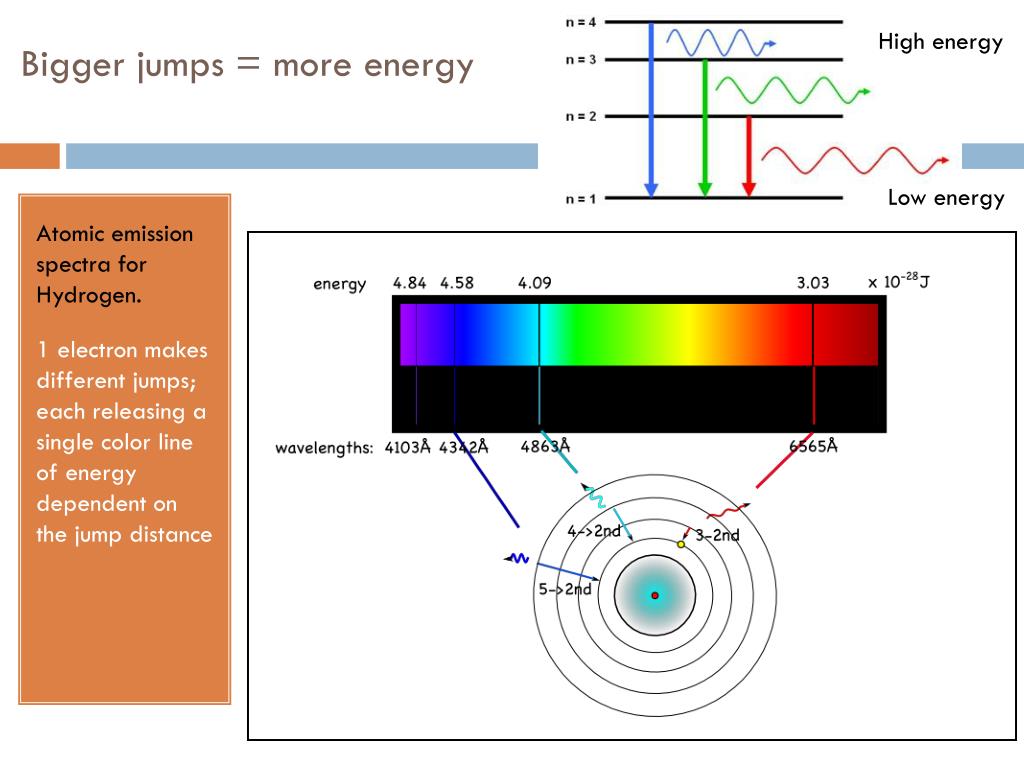
#Atomic emission spectra series
electric lamp with tungsten filament or energy saving light bulb.You will be using a hand spectroscope to observe the difference between a continuous emission spectrum produced by a fluorescent and a line spectrum produced by the excitation of electrons in various elements. This experiment is designed to give you a qualitative introduction to the spectra emitted by some s-block elements when their atoms are excited by heating in a Bunsen flame.

